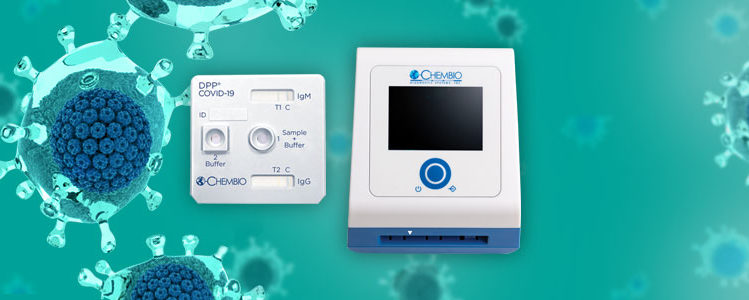
Hauppauge-based Chembio Diagnostics said it is launching a 15-minute COVID-19 test in the United States with shipments slated to begin this month.
The company, which specializes in making tests for infectious diseases, in a press release said it is launching its rapid DPP COVID-19 serological point-of-care test that detects antibodies related to the disease. Results can be obtained within 15 minutes after pricking a finger by using analyzers that are produced by Chembio in Germany.
“Our measured approach has positioned us to offer a viable and sustainable long-term solution for clinicians,” CEO Richard Eberly said. “We expect to begin shipping product in April 2020.”
The test can determine whether patients have been exposed to the novel coronavirus, even if they have no symptoms, whether a person is infectious and could possibly transmit the infection to another person. And it can determine whether a person had been infected but is no longer able to transmit the virus.
Eberly added the company will continue to work with its partner LumiraDx to provide “COVID-19 tests with the ability to scale based upon market demand.”
“By joining forces and bringing together the best of these two companies, we believe we will become the chosen approach for the detection and monitoring of the COVID-19 virus, which has become a worldwide pandemic,” LuminaDx CEO Ron Zwanziger said earlier.
The company said the test will be distributed as authorized by the United States Food and Drug Administration’s notification process under the public health emergency guidance issued on March 16.
“This is another example of Chembio’s ability to respond in an expeditious manner to global pandemics with differentiated solutions,” Chembio Director Gail Page said, citing tests for Zika and Ebola.
Other companies also have developed and are beginning to use 15-minute tests, as quick detection becomes a priority in order to identify infection and stem its spread.